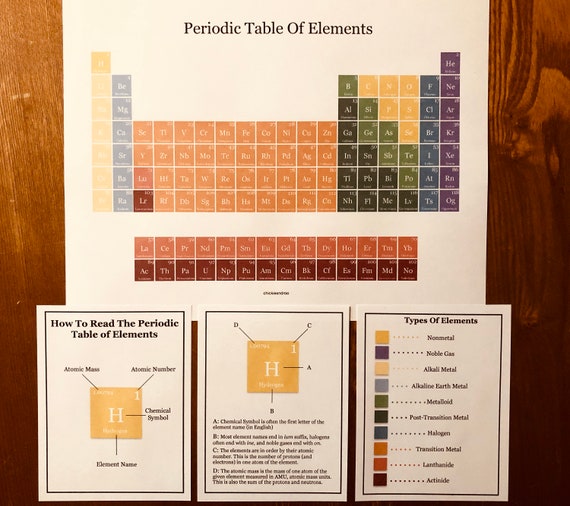
Students should be familiar with the definitions of an element and an atom the basic structure of the atom, including valence electrons and the physical and chemical properties of elements. Compare and contrast multiple versions of the periodic table. sodium hydroxide solid soluble solution speed steel Step structure substance sulfate.phase changes and physical/chemical changes as well as tie into our next. Chemical reactions (their properties, types, flow conditions, etc. Periodic Tables Online (pdf) - A worksheet I use to review the basics of the. Investigate repeating patterns and predict periodicity for atomic radius, electronegativity, and ionization energy. An effective method of introducing the periodic table as a crossword puzzle at the high school level. Limestone or calcium carbonate (caco3) decomposes when heated to a high temperature.Organize the element cards based on the patterns of the elements’ physical and chemical properties.Beginning with hydrogen in the top left of the table, atomic numbers increase from left to right and top to bottom. 
Hydrogen (H) is the first element with an atomic number of 1. PS1.A: Structure and Properties of Matter You may have noticed that the periodic table is also arranged in order of increasing atomic number. Like a compass for a navigator, it guides teachers and learners through the world of matter. Digital resources may include a teacher manual and student guide, pre-lab activities and setup videos, phenomenon videos, simulations, and post-lab analysis and assessments. In the chemistry classroom, the periodic table holds a special place of honor. Includes 1-year access to digital resources that support 3-dimensional instruction for NGSS. They then compare their models to the periodic table and 3 alternative periodic tables with the goal of answering the driving question, “What patterns can be identified in the design of the periodic table, and how can those patterns be used to make predictions?” Elements having the same number of electrons in their outermost shell are placed in vertical columns called groups. In this lab, students seek to understand this phenomenon by constructing models of the elements based on their chemical and physical characteristics. This modern periodic table lists elements according to their atomic number, if they were arranged according to atomic mass potassium and argon would be the wrong way round. Astrobiologists have theorized that life on other planets might be silicon based as opposed to the carbon-based life that exists on Earth.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
